Puros® Dermis Allograft Tissue Matrix

Puros® Dermis Allograft Tissue Matrix The Natural Choice for Soft-Tissue Augmentation
- Puros Dermis Allograft Tissue Matrix is a high-quality regenerative material, exhibiting multidirectional strength2 and adaptability to surface contours5
- Rehydrates quickly, no refrigeration required, packaged sterile and no residual antibiotics4
Significantly Better Handling* with Tutoplast Processing
*Ability to hydrate the allograft, ability to handle and manipulate the hydrated product, adaptability to contours, structural integrity during manipulation.
Puros Dermis Allograft Tissue Matrix Features & Benefits
1. Versatile Soft-Tissue Augmentation
- Utilized for both horizontal and vertical soft-tissue augmentation and guided tissue regeneration procedures1,2
- Reduces morbidity and saves valuable chair time by eliminating the need to harvest an autogenous graft3
2. Natural Regeneration
- Retains the natural collagen matrix, content, and mechanical properties of native dermis4
- Provides a healing environment and acts as a guide for the regeneration of vital soft-tissue1,2
3. Trusted source
- Sterilized using the proprietary Tutoplast® process
- Excellent handling, rapid hydration4
Puros Dermis Allograft Tissue Matrix Variety of Clinical Advantages
- Successful clinical history in other surgical applications5
- Petrungaro found enhanced volume by maintaining space to allow for angiogenesis, tissue remodeling, and increased volume of attached gingiva and connective tissue1,2
- Biocompatible and absorbable; assimilates into the body’s normal tissue healing process at the histological level3,4
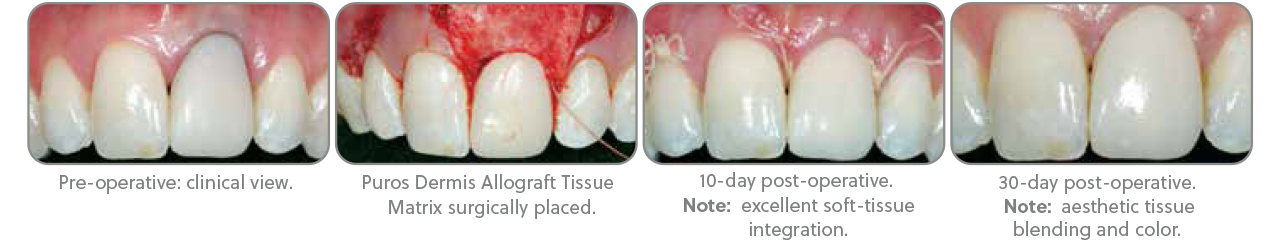
Clinical photographs ©2012 Paul S. Petrungaro†, DDS, MS. All rights reserved. Individual results may vary.
Puros Allograft Block Unique Tutoplast Process
In 1969 the Tutoplast Tissue Sterilization Process was developed to sterilize and preserve tissue for implantation. More than 11 million implants have been sterilized through the Tutoplast Process with zero confirmed incidence of implant-associated infection.17
The Benefits of the Multi-Step Tutoplast Process
For allograft bone grafts, the process preserves the valuable bone mineral, collagen matrix, and tissue integrity 18 while inactivating pathogens and gently removing unwanted materials, such as cells, antigens, and viruses 17 – resulting in predictable, reliable, sterile, and safe tissue.17
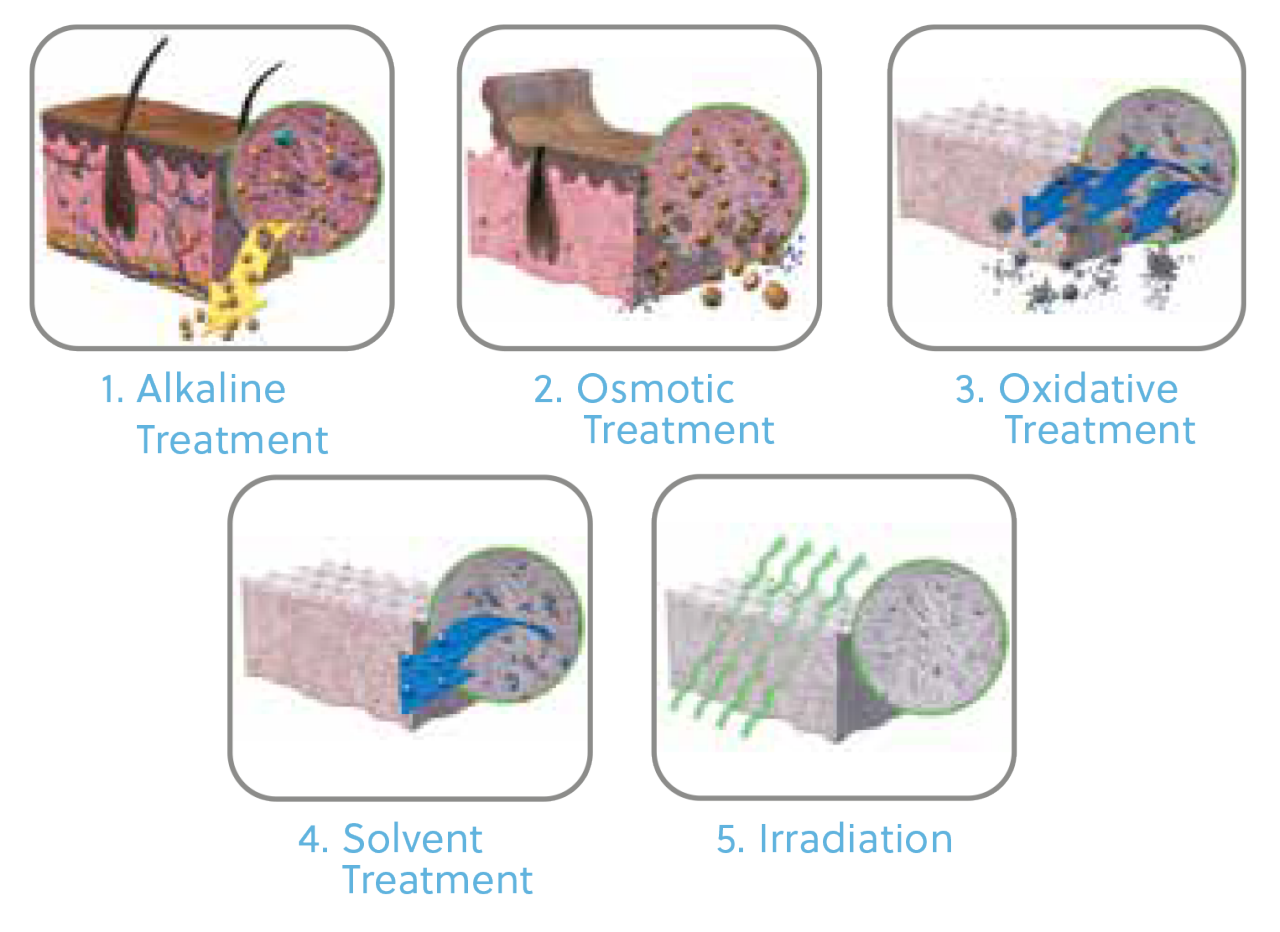
*Images depict dermal processing