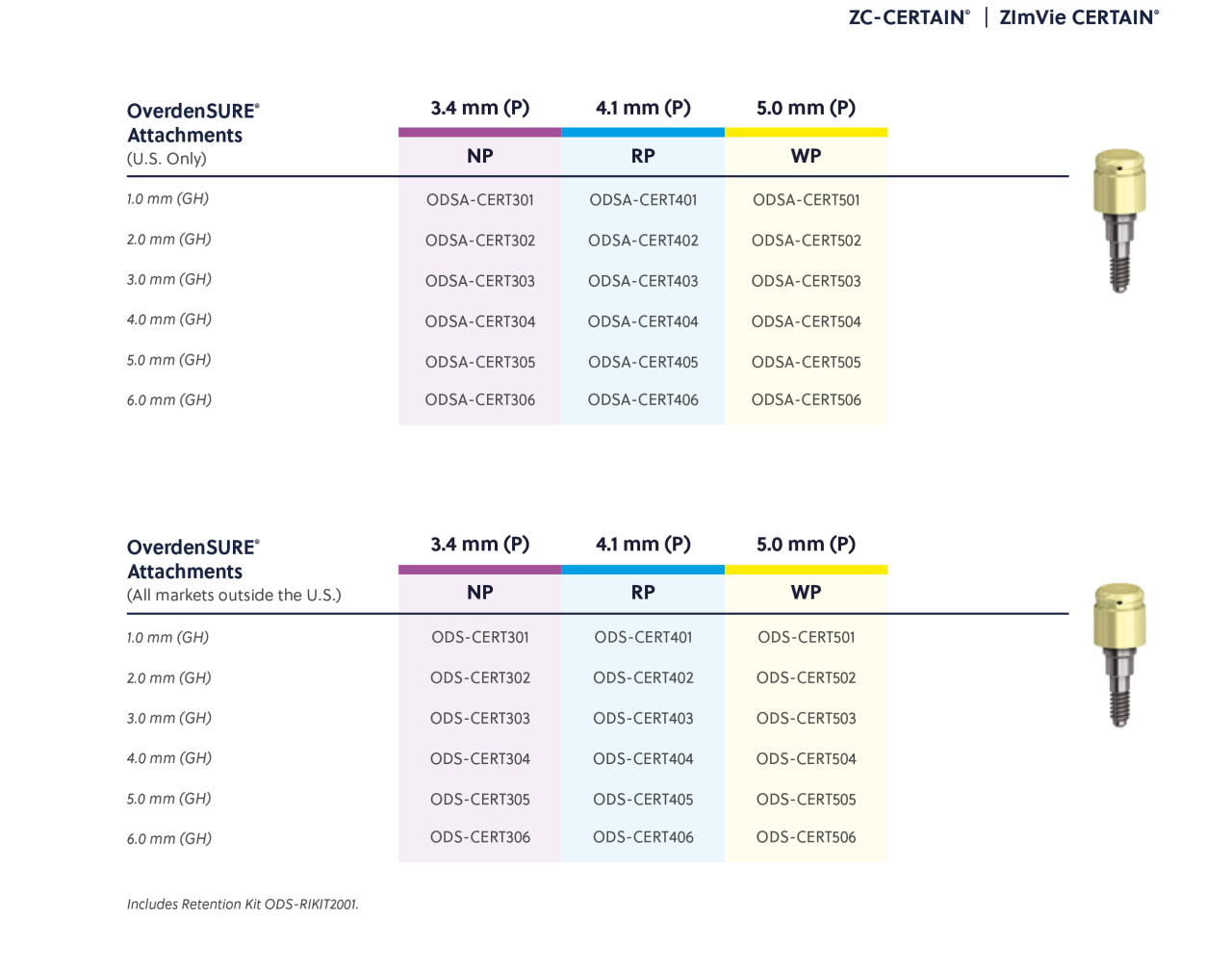
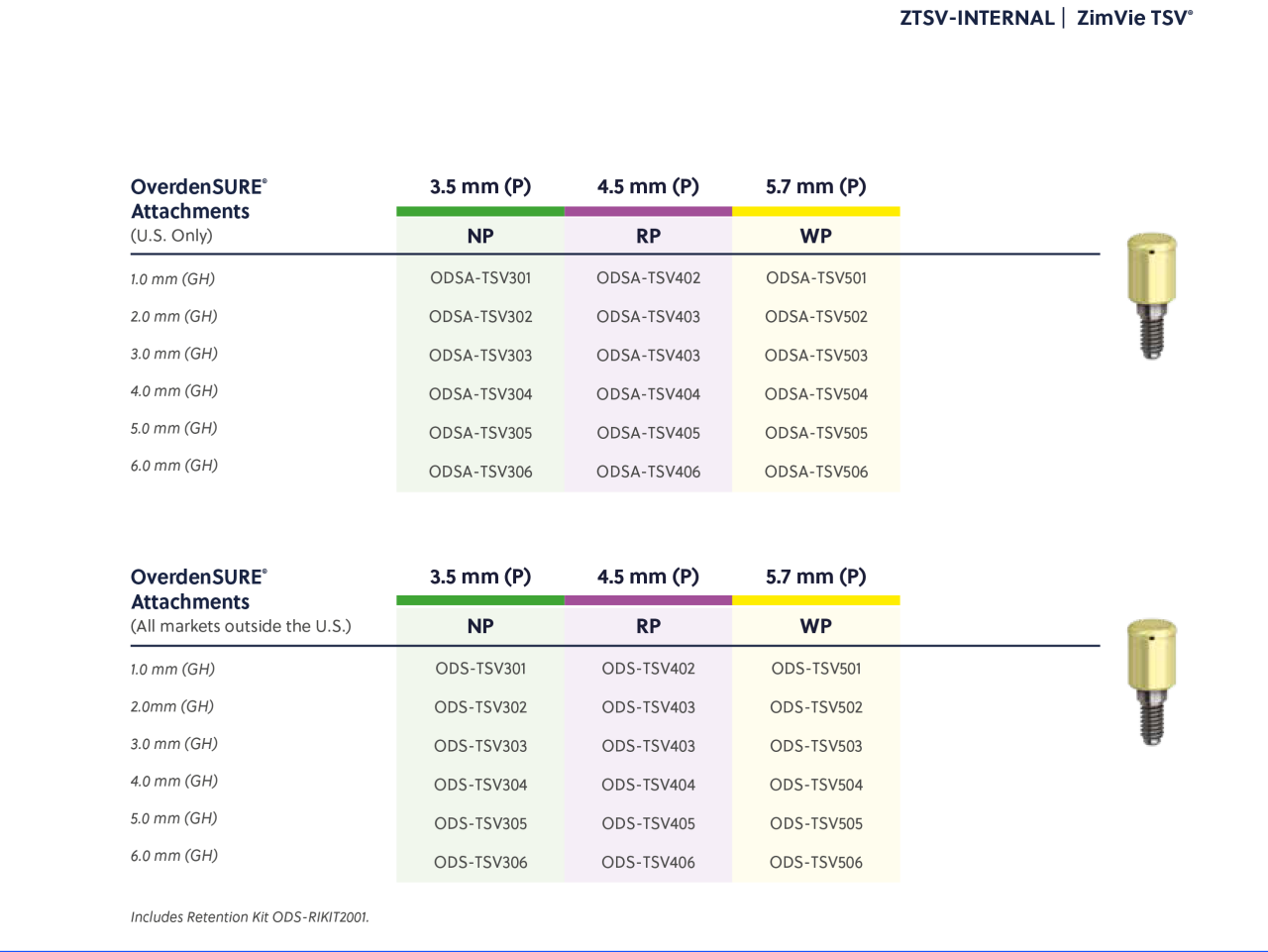
OverdenSURE®
Removable Attachment System
Compatible with the market's leading implant systems!
Providing increased prosthetic stability and retention for patients wearing conventional dentures.

The Clinical Challenge
It is estimated that 10% of the world’s population is partially edentulous or edentulous1.
Despite advances, soft-tissue supported dentures continue to be a source of:
Chronic residual ridge resorption
Decreased masticatory function
A loose or poor fit
Phonetic challenges
Facial collapse

Benefits
Together with our dental implant solutions, OverdenSURE Abutments are an ideal option for overdenture restorations, providing increased prosthetic stability and retention for patients wearing conventional dentures.
Zirconium Nitride abutment coating for excellent aesthetics and function.
Wider range of abutment sizes for increased restorative flexibility.
Fully bio-compatible material exhibits increased hardness and wear resistance1,2.
Reduced bacterial adhesion3,4,5,6,7.
Enhanced lubricity compared to the traditionally used Titanium Nitride1.
Classic Housing Cap and Retention Insert design addresses problems such as limited interocclusal space through the OverdenSURE Abutment’s low restorative height.
Features
Good just got better with ZimVie's removable attachment system, OverdenSURE!

OverdenSURE Removable Attachment System
Classic Housing Cap and Retention Insert design
Low restorative height for limited interocclusal spaces
Total vertical height of 3.17 mm
(Including abutment crown, Retention Insert and Housing Cap)
OverdenSURE Pivoting Technology
Designed to swivel inside the fixed Housing Cap for easy seating – even when the implants are placed off axis
Allows patients to place dentures when not fully aligned with the abutments
Retention Inserts have been designed to swivel inside the fixed Housing Cap enabling the easy seating of the removable prosthesis even when:
Implants are placed off axis.
The denture is not fully aligned with the abutments.

Standard Retention Inserts
Standard Retention Inserts provide angle correction for implants up to 10º of divergence off axis, and a total of 20º of divergence between two implants.

Extended Retention Inserts
Extended Retention Inserts accommodate for up to 20º of implant divergence off axis, and a total of 40º of divergence between two implants.